PeronLab@NYU
Probing the cortical circuit basis of perception

Probing the cortical circuit basis of perception

Cortical activity underpins perception. This activity is often sparse, with a minority of neurons responding to specific stimuli. How do sparse groups of neurons influence perception and behavior? How does cortical computation impact perception? We use the mouse whisker system to tackle these questions.

Large-scale recording of cortical activity in animals performing controlled behaviors allows us to identify populations that may underpin perception. Such correlative experiments are an essential prerequisite to experiments that probe circuit computation and the perceptual role of specific populations.

Recurrence is a defining feature of cortical circuitry, and is believed to underpin many computations. We use perturbation in conjunction with network models to probe how recurrence among specific populations influences cortical computation.

Identifying the cortical populations that correlate with perception is not enough to establish causality. We use focal perturbation to understand how specific populations of neurons contribute to behavior.

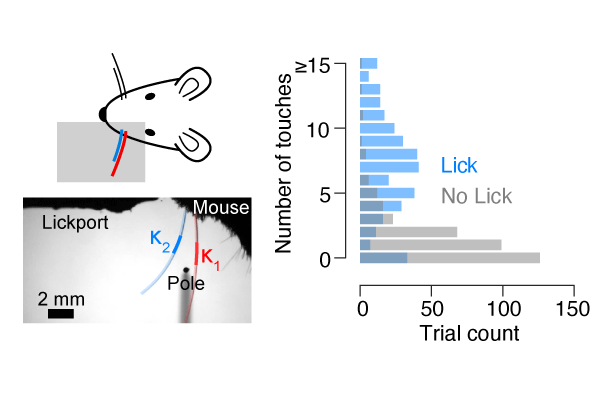
Combining high speed videography with modern image processing techniques allows for precise measurement of the behavioral state of the animal. Tasks are designed so that even subtle changes in the animal's behavior can be detected.
Two-photon microscopy using modern calcium indicators allows us to record the activity of thousands of cortical neurons during behavior. Neurons can be tracked over months, allowing us to study how cortical dynamics evolve over time.

Our experiments depend on rapid turnaround of terabyte-scale datasets. We are developing pipelines to process data acquired during calcium imaging and behavioral videography, and to relate neural activity to behavior.

Neurons encoding particular features are often intermingled with other neuron types. Approaches like multiphoton ablation, microlesions, and two-photon optogenetics allow us to test the hypotheses originating from our large scale mapping experiments.

Ahmed A, Voelcker B, Peron SP
2023, BioRxiv
Pancholi R, Sun-Yan A, Laughton M, Peron SP
2023, BioRxiv
Ryan L, Sun-Yan A, Laughton M, Peron SP
2023, Cell Reports
Pancholi R, Ryan L, Peron SP
2023, Nature Communications
Pancholi R, Sun-Yan A, Peron SP
2023, Current Biology
Ryan L, Laughton M, Sun-Yan A, Costello S, Pancholi R, Peron SP
2022, eNeuro
Voelcker B, Pancholi R, Peron SP
2022, Nature Communications
Peron SP, Pancholi R, Voelcker B, Wittenbach JD, Olafsdottier FH, Freeman J, Svoboda K
2020, Nature
Peron SP, Chen TW, Svoboda K
2015, Curr. Opinion Neurobiology
Peron SP, Freeman J, Iyer V, Guo C, Svoboda K
2015, Neuron

Simon earned his PhD with Fabrizio Gabbiani at Baylor College of Medicine, studying single neuron computation in the context of insect vision. He did his postdoctoral work with Karel Svoboda at Janelia Farm, working on mechanisms of cortical processing in the behaving mouse using two-photon microscopy.

Ravi joined the lab in 2018. Ravi employs optical microstimulation to study the neural basis of perception.

Lauren joined the lab in 2019. Lauren's work focuses on the role sensory cortex plays in behavior.

Alisha joined the lab in 2022. Alisha is interested in representational drift in vibrissal sensorimotor cortices.

Maya joined the lab in 2021. Maya is our master animal wrangler.

Andrew joined the lab in 2021.
We are actively recruiting post-docs and PhD candidates. If you are interested in our ongoing projects, please get in touch!
Cortical activity has long been known to contribute to perception: nearly a century ago, Penfield and colleagues demonstrated that direct stimulation of somatosensory cortex could evoke natural touch sensations. How do the neurons in the mouse vibrissal cortical areas influence the perception of touch? We recently demonstrated that a single cortical barrel containing approximately 10,000 neurons is crucial to object localization but not detection behaviors. We now seek to use cellular-resolution perturbation to determine if much smaller populations - 50 to 100 neurons - actually underpin localization behaviors. We seek candidates interested in how cortical activity results in specific percepts.
Cortex performs a range of computations that ultimately influence behavior. Understanding these computations requires careful measurement of neural activity combined with precise perturbation of circuit elements. We use a range of techniques, including volumetric calcium imaging, multiphoton ablation, and optogenetic approaches to asses how barrel cortex transforms sensory inputs. Building on recent work that revealed increasing receptive field complexity in the most superficial layers of barrel cortex, we seek candidates interested in designing experiments to probe the way in which specific functional classes of excitatory pyramidal neurons interact to produce these receptive fields.
Barrel cortex contains groups of touch-sensitive neurons that are recurrently coupled, allowing for computations such as amplification and pattern completion. How do these recurrent networks emerge, and what rules govern their formation? We recently developed an optical microstimulation paradigm that allows us to generate such recurrent networks artificially in mice that then use the evoked activity to drive behavior. We seek candidates interested in building on this work to probe the rules governing recurrent network plasticity in vivo.
Peron Lab
Center for Neural Science
New York University
4 Washington Place, Rm. 809
New York, NY 10003